Quest for the right Drug
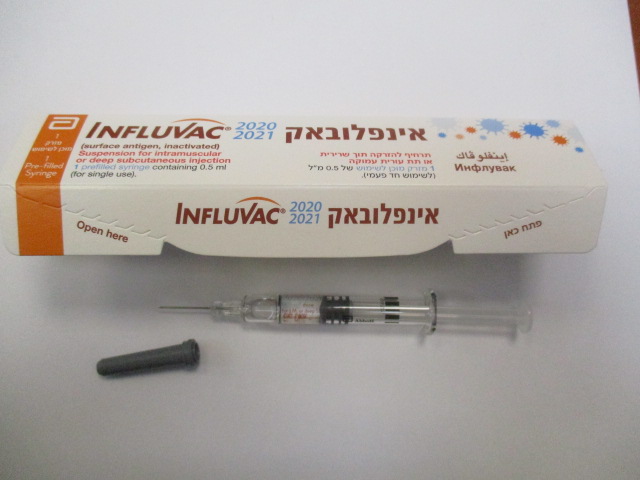
אינפלובאק INFLUVAC (A/CAMBODIA/E0826360/2020 (H3N2)-LIKE STRAIN, A/GUANGDONG-MAONAN/SWL1536/2019 (H1N1) PDM09-LIKE VIRUS, A/HONG KONG/2671/2019 (H3N2)-LIKE VIRUS, A/VICTORIA/2570/2019 (H1N1)PDM09-LIKE STRAIN, B/WASHINGTON/02/2019 (B/VICTORIA LINEAGE)-LIKE VIRUS)
תרופה במרשם
תרופה בסל
נרקוטיקה
ציטוטוקסיקה
צורת מתן:
תוך-שרירי, תת-עורי עמוק : I.M, DEEP S.C.
צורת מינון:
תרחיף להזרקה : SUSPENSION FOR INJECTION
עלון לרופא
מינוניםPosology התוויות
Indications תופעות לוואי
Adverse reactions התוויות נגד
Contraindications אינטראקציות
Interactions מינון יתר
Overdose הריון/הנקה
Pregnancy & Lactation אוכלוסיות מיוחדות
Special populations תכונות פרמקולוגיות
Pharmacological properties מידע רוקחי
Pharmaceutical particulars אזהרת שימוש
Special Warning עלון לרופא
Physicians Leaflet
Special Warning : אזהרת שימוש
4.4. Special warnings and precautions for use As with all injectable vaccines, appropriate medical treatment and supervision should always be readily available in case of an anaphylactic event following the administration of the vaccine. Influvac should under no circumstances be administered intravascularly. As with other vaccines administered intramuscularly, Influvac should be given with caution to individuals with thrombocytopenia or any coagulation disorder since bleeding may occur following an intramuscular administration to these subjects. Anxiety-related reactions, including vasovagal reactions (syncope), hyperventilation or stress-related reactions can occur following, or even before, any vaccination as a psychogenic response to the needle injection. This can be accompanied by several neurological signs such as transient visual disturbance, paraesthesia and tonic-clonic limb movements during recovery. It is important that procedures are in place to avoid injury from faints. Influvac is not effective against all possible strains of influenza virus. Influvac is intended to provide protection against those strains of virus from which the vaccine is prepared and to closely related strains. As with any vaccine, a protective immune response may not be elicited in all vaccinees. Antibody response in patients with endogenous or iatrogenic immunosuppression may be insufficient. Interference with serological testing: see section 4.5. This medicine contains less than 1 mmol sodium (23 mg) per dose, i.e. essentially ‘sodium- free’. This medicine contains potassium, less than 1 mmol (39 mg) per dose, i.e. essentially “potassium- free”.
Effects on Driving

שימוש לפי פנקס קופ''ח כללית 1994
Vaccination is recommended in the following high risk categories: congenital or acquired heart disease, chronic renal disease, chronic bronchopulmnary disease, diabetes mellitus & other metabolic diseases, chronic severe anemia, immunocompromised persons, age over 65 years
תאריך הכללה מקורי בסל
01/01/1995
הגבלות
תרופה שאושרה לשימוש כללי בקופ'ח
מידע נוסף
