Quest for the right Drug
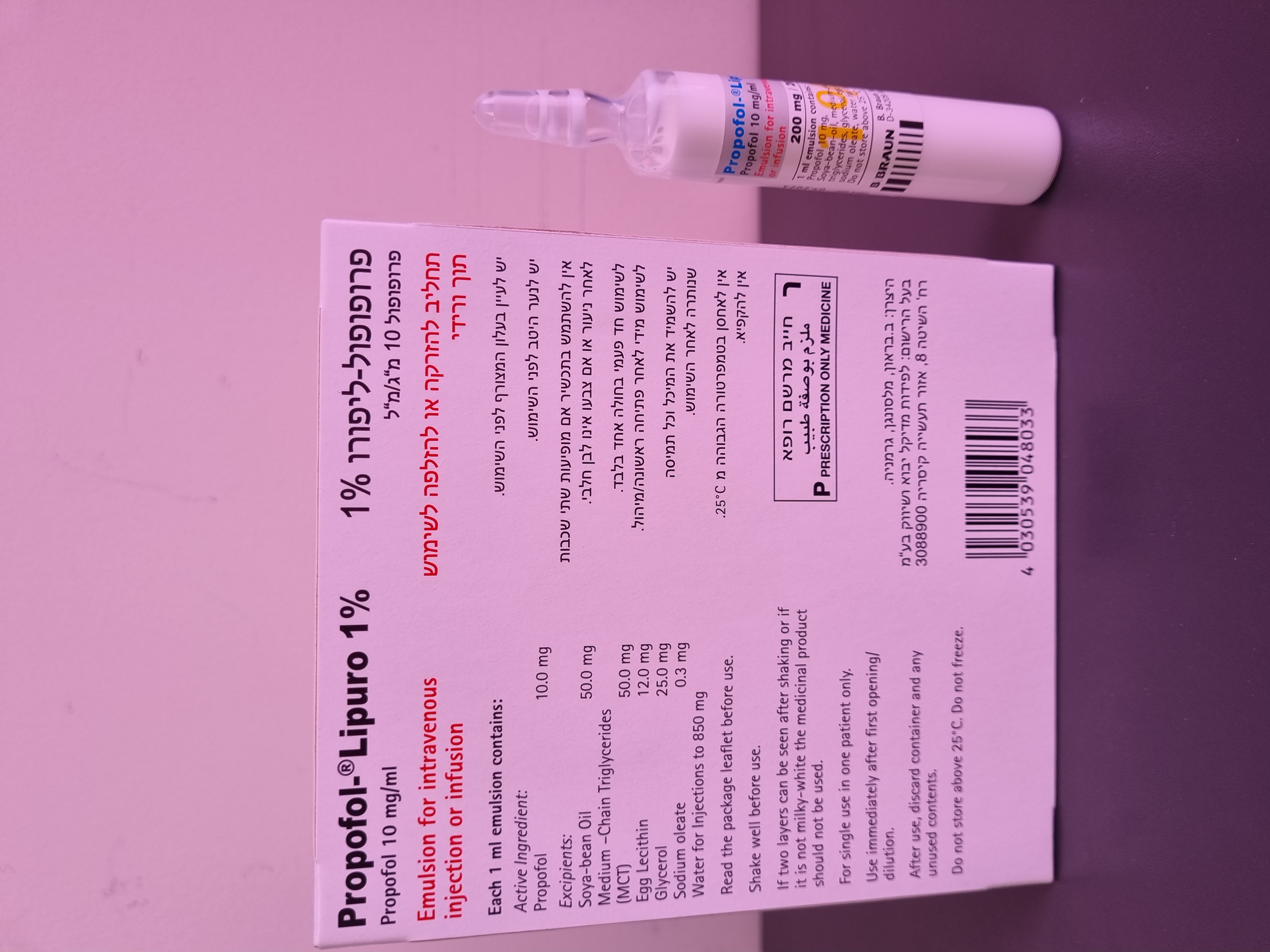
פרופופול ליפורו % 1 PROPOFOL - LIPURO 1 % (PROPOFOL)
תרופה במרשם
תרופה בסל
נרקוטיקה
ציטוטוקסיקה
צורת מתן:
תוך-ורידי : I.V
צורת מינון:
תחליב להזרקה או אינפוזיה : EMULSION FOR INJECTION OR INFUSION
עלון לרופא
מינוניםPosology התוויות
Indications תופעות לוואי
Adverse reactions התוויות נגד
Contraindications אינטראקציות
Interactions מינון יתר
Overdose הריון/הנקה
Pregnancy & Lactation אוכלוסיות מיוחדות
Special populations תכונות פרמקולוגיות
Pharmacological properties מידע רוקחי
Pharmaceutical particulars אזהרת שימוש
Special Warning עלון לרופא
Physicians Leaflet
Posology : מינונים
4.2 Posology and Method of Administration General instructions Propofol-Lipuro 1% must only be given in hospitals or adequately equipped day therapy units by phy- sicians trained in anaesthesia or in the care of patients in intensive care. Circulatory and respiratory functions should be constantly monitored (e.g. ECG, pulse-oxymeter) and facilities for maintenance of patent airways, artificial ventilation, and other resuscitation facilities should be immediately avail- able at all times. For sedation during surgical or diagnostic procedures Propofol-Lipuro 1% should not be given by the same person that carries out the surgical or diagnostic procedure. Supplementary analgesic medicinal products are generally required in addition to Propofol-Lipuro 1% Posology Propofol-Lipuro 1% is given intravenously. The dosage is adjusted individually according to the pa- tient’s response. ● General anaesthesia in adults Induction of anaesthesia: For induction of anaesthesia Propofol-Lipuro 1% should be titrated (20 – 40 mg of propofol every 10 seconds) against the patient’s response until the clinical signs show the onset of anaesthesia. Most adult patients younger than 55 years are likely to require 1.5 to 2.5 mg/kg body weight. In patients over this age and in patients of ASA grades III and IV, especially those with impaired car- diac function, the dosage requirements will be less and the total dose of Propofol-Lipuro 1% may be reduced to a minimum of 1 mg/kg body weight. In these patients lower rates of administration should be applied (approximately 2 ml, corresponding to 20 mg every 10 seconds). Maintenance of anaesthesia: Anaesthesia can be maintained by administering Propofol-Lipuro 1% either by continuous infusion or by repeat bolus injections. If a technique involving repeat bolus injections is used, increments of 25 mg (2.5 ml Propofol-Lipuro 1% to 50 mg (5.0 ml Propofol-Lipuro 1% may be given according to clinical requirements. For maintenance of anaesthesia by continuous infusion the dosage requirements usually are in the range of 4 – 12 mg/kg body weight/h. In elderly patients, in patients of poor general condition, in patients of ASA grades III and IV and in hypovolaemic patients the dosage may be reduced further depending on the severity of the patient’s condition and on the performed anaesthetic technique. ● General anaesthesia in children over 1 month of age Induction of anaesthesia: For induction of anaesthesia Propofol-Lipuro 1% should be slowly titrated against the patient’s re- sponse until the clinical signs show the onset of anaesthesia. The dosage should be adjusted according to age and/or body weight. Most patients over 8 years of age require approximately 2.5 mg/kg body weight of propofol for induc- tion of anaesthesia. In younger children, especially between the age of 1 month and 3 years, dose re- quirements may be higher (2.5 – 4 mg/kg body weight). Maintenance of general anaesthesia: Anaesthesia can be maintained by administering Propofol-Lipuro 1% by infusion or repeated bolus injection to maintain the depth of anaesthesia required. The required rate of administration varies con- siderably between patients but rates in the region of 9 – 15 mg/kg/h usually achieve satisfactory an- aesthesia. In younger children, especially between the age of 1 month and 3 years, dose requirements may be higher. For ASA III and IV patients lower doses are recommended (see also section 4.4) ● Sedation of ventilated patients in the Intensive Care Unit For sedation during intensive care it is advised that propofol should be administered by continuous in- fusion. The infusion rate should be determined by the desired depth of sedation. In most patients suf- ficient sedation can be obtained with a dosage of 0.3 - 4 mg/kg/h of propofol (see also section 4.4). Propofol is not indicated for sedation in intensive care of patients of 16 years of age or younger (see section 4.3). Administration of propofol by Target Controlled Infusion (TCI) system is not advised for sedation in the intensive care unit. ● Sedation for diagnostic and surgical procedures in adults To provide sedation during surgical and diagnostic procedures, doses and administration rates should be adjusted according to the clinical response. Most patients will require 0.5 – 1 mg/kg body weight over 1 to 5 minutes for onset of sedation. Maintenance of sedation may be accomplished by titrating Propofol-Lipuro 1% infusion to the desired level of sedation. Most patients will require 1.5 – 4.5 mg/kg body weight/h. The infusion may be supplemented by bolus administration of 10 – 20 mg (1 – 2 ml Propofol-Lipuro 1% if a rapid increase of the depth of sedation is required. In patients older than 55 years and in patients of ASA grades III and IV lower doses of Propofol- Lipuro 1% may be required and the rate of administration may need to be reduced. ● Sedation for diagnostic and surgical procedures in children over 1 month of age Doses and administration rates should be adjusted according to the required depth of sedation and the clinical response. Most paediatric patients require 1 – 2 mg/kg body weight of propofol for onset of sedation. Maintenance of sedation may be accomplished by titrating Propofol-Lipuro 1% as infusion to the desired level of sedation. Most patients require 1.5 – 9 mg/kg/h of propofol. The infusion may be supplemented by bolus administration of up to 1 mg/kg b.w. if a rapid increase of depth of sedation is required. In ASA III and IV patients lower doses may be required. Method and duration of administration ● Method of administration Intravenous use Propofol-Lipuro 1% is administered intravenously by injection or continuous infusion either undilut- ed or diluted with 5 % w/v glucose solution or 0.9 % w/v sodium chloride solution as well as in a 0.18 % w/v sodium chloride and 4 % w/v glucose (see also section 6.6). Containers should be shaken before use. Before use, the neck of the ampoule or the surface of the rubber stopper of the vial should be cleaned with medicinal alcohol (spray or swabs). After use, tapped containers must be discarded. Propofol-Lipuro 10 mg/ml contains no antimicrobial preservatives and supports growth of microorganisms. Therefore, Propofol-Lipuro 10 mg/ml is to be drawn up aseptically into a sterile syringe or an infusion set immediately after opening the ampoule or breaking the vial seal. Administration must commence without delay. Asepsis must be maintained for both Propofol-Lipuro 10 mg/ml and the infusion equipment throughout the infusion period. Any medicinal products or fluids added to a running Propofol-Lipuro 10 mg/ml infusion must be administered close to the cannula site. If infusion sets with filters are to be used, these must be lipid- permeable. Propofol-Lipuro 1 % (10 mg/ml) must not be administered via infusion sets with microbi- ological filters. The contents of one ampoule or one vial of Propofol-Lipuro 10 mg/ml and any syringe containing Propofol-Lipuro 10 mg/ml are for single use in one patient. Infusion of undiluted Propofol-Lipuro 1% When administering Propofol-Lipuro 1% by continuous infusion, it is recommended that burettes, drop counters, syringe pumps or volumetric infusion pumps, should always be used to control the in- fusion rates. As established for the parenteral administration of all kinds of fat emulsions, the duration of continuous infusion of Propofol-Lipuro 1 % (10 mg/ml) from one infusion system must not exceed 12 hours. The infusion line and the reservoir of Propofol-Lipuro 1 % (10 mg/ml) must be discarded and replaced after 12 hours at the latest. Any portion of Propofol-Lipuro 1 % (10 mg/ml) remaining after the end of infusion or after replacement of the infusion system must be discarded. Infusion of diluted Propofol-Lipuro 1% For administering infusion of diluted Propofol-Lipuro 1%, burettes, drop counters, syringe pumps, or volumetric infusion pumps should always be used to control infusion rates and to avoid the risk of ac- cidentally uncontrolled infusion of large volumes of diluted Propofol-Lipuro 1%. The maximum dilution must not exceed 1 part of Propofol-Lipuro10 mg/ml with 4 parts of 5 % w/v glucose solution or 0.9 % w/v sodium chloride solution, or 0.18 % w/v sodium chloride and 4 % w/v glucose solution (minimum concentration 2 mg propofol/ml). The mixture should be prepared asepti- cally immediately prior to administration and must be used within 6 hours of preparation. In order to reduce pain on initial injection, Propofol-Lipuro 1% may be mixed with preservative-free lidocaine injection 1 % (mix 20 parts of Propofol-Lipuro 1% with up to 1 part of lidocaine injection 1%). Before giving the muscle relaxants atracurium or mivacurium subsequent to Propofol-Lipuro 10 mg/ml through the same intravenous line, it is recommended that the line be rinsed prior to admin- istration. Propofol may also be used by Target Controlled Infusion. Due to the different algorithms available on the market for dosage recommendations please refer to the instructions for use leaflet of the device manufacturer. ● Duration of administration Propofol-Lipuro 1% can be administered for a maximum period of 7 days.

שימוש לפי פנקס קופ''ח כללית 1994
לא צוין
תאריך הכללה מקורי בסל
לא צוין
הגבלות
לא צוין
מידע נוסף
