Quest for the right Drug
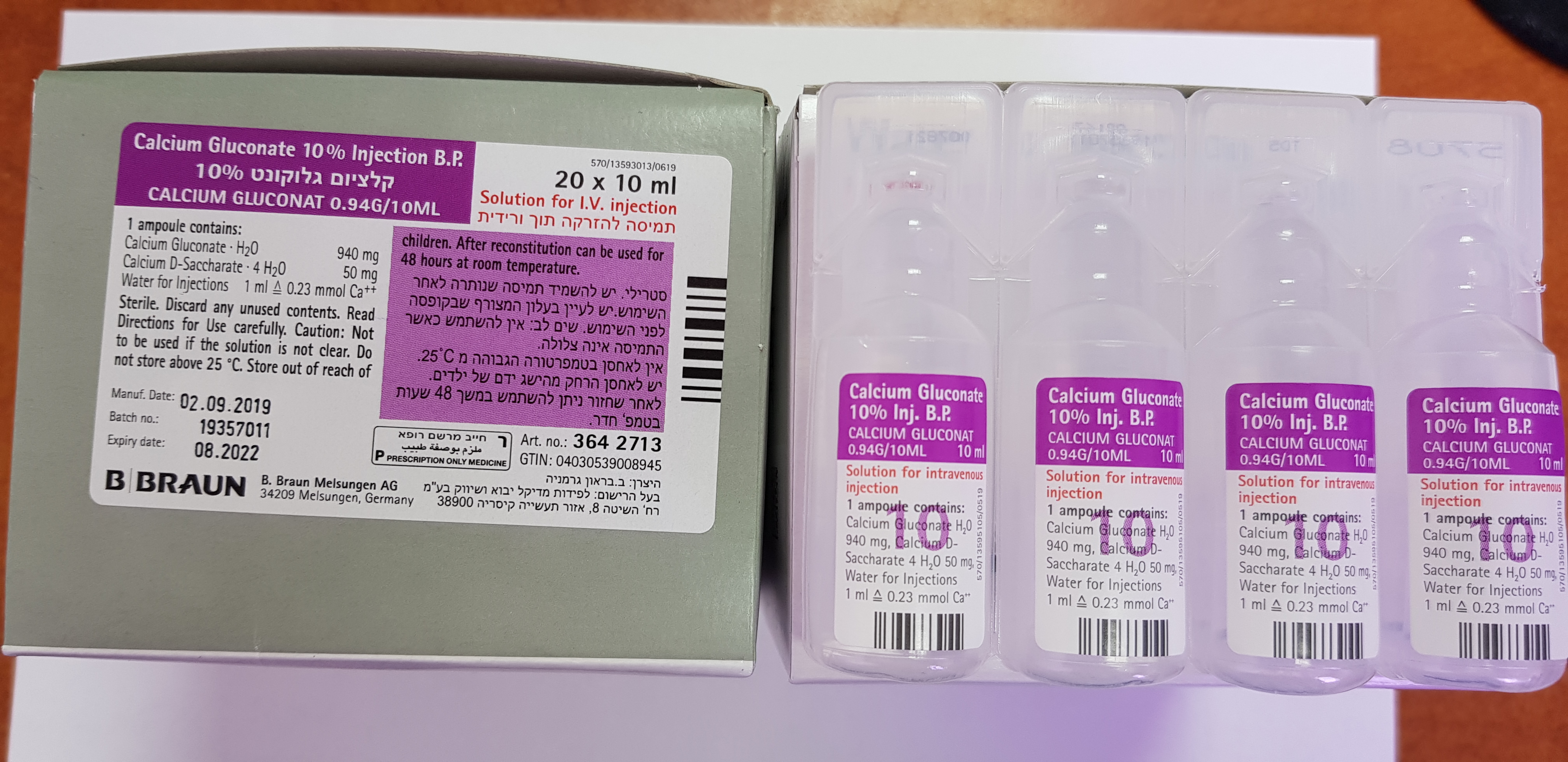
קלציום גלוקונט % 10 CALCIUM GLUCONATE 10 % INJECTION B.P. (CALCIUM GLUCONATE)
תרופה במרשם
תרופה בסל
נרקוטיקה
ציטוטוקסיקה
צורת מתן:
תוך-ורידי : I.V
צורת מינון:
תמיסה להזרקה : SOLUTION FOR INJECTION
עלון לרופא
מינוניםPosology התוויות
Indications תופעות לוואי
Adverse reactions התוויות נגד
Contraindications אינטראקציות
Interactions מינון יתר
Overdose הריון/הנקה
Pregnancy & Lactation אוכלוסיות מיוחדות
Special populations תכונות פרמקולוגיות
Pharmacological properties מידע רוקחי
Pharmaceutical particulars אזהרת שימוש
Special Warning עלון לרופא
Physicians Leaflet
Pharmaceutical particulars : מידע רוקחי
6 PHARMACEUTICAL PARTICULARS 6.1 List of excipients Calcium-D-saccharate tetrahydrate Water for injections 6.2 Incompatibilities Calcium salts can form complexes with many drugs and this may result in a precipitate. Calcium salts are incompatible with oxidising agents, citrates, soluble carbonates, bicarbonates, oxalates, phosphates, tartrates and sulphates. Physical incompatibility has also been reported with amphotericin, cephalothin sodium, ceftriaxone (see section 4.4), cephazolin sodium, cephamandole nafate, novobiocin sodium, dobutamine hydrochloride, prochlorperazine and tetracyclines. This medicinal product must not be mixed with other medicinal products except those mentioned in section 6.6 or unless compatibility has been satisfactorily demonstrated. 6.3 Shelf life Unopened The expiry date of the product is indicated on the packaging materials. After dilution When diluted to 10mg/ml, according to directions, with the recommended infusion fluids (i.e. sodium chloride 9mg/ml (0.9%) solution for injection or 50mg/ml (5%) glucose solution for injection) physical in-use stability has been demonstrated for 48 hours at room temperature. From a microbiological point of view, the diluted product should be used immediately. If not used immediately, in-use storage times and conditions prior to use are the responsibility of the user and would normally not be longer than 24 hours at 2 to 8ºC, unless dilution has taken place in controlled and validated aseptic conditions. 6.4 Special precautions for storage Do not store above 25°C. 6.5 Nature and contents of container 10ml LDPE ampoules packed in cardboard box. Pack size: 20 ampoules. Not all pack sizes may be marketed. 6.6 Special precautions for disposal Disposal No special requirements for disposal. Handling The product is intended for single use only. Discard any unused solution. The medicinal product should be visually inspected for particulate matter, discoloration and the integrity of the container prior to use. The solution should only be used if it is clear, colourless to light brownish, practically free from visible particles and the container is undamaged. Dilution For intravenous infusion, Calcium Gluconate 10 % Injection B.P. may be diluted 1:10 to a concentration of 10mg/ml with the following two infusion fluids: sodium chloride 9mg/ml (0.9%) solution for injection or 50mg/ml (5%) glucose solution for injection. When diluted with these recommended infusion fluids, the resulting solutions are intended for immediate single use. Dilution should be performed under controlled and validated aseptic conditions. After mixing, the container should be gently agitated to ensure homogeneity.

שימוש לפי פנקס קופ''ח כללית 1994
Calcium supplement
תאריך הכללה מקורי בסל
01/01/1995
הגבלות
תרופה שאושרה לשימוש כללי בקופ'ח
מידע נוסף
