Quest for the right Drug
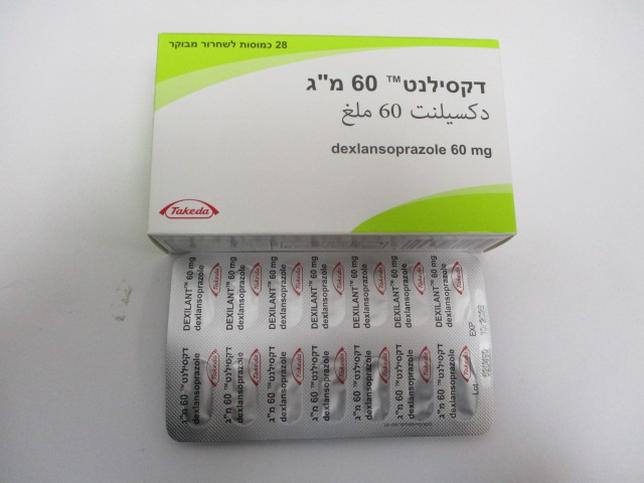
דקסילנט 60 מ"ג DEXILANT 60 MG (DEXLANSOPRAZOLE)
תרופה במרשם
תרופה בסל
נרקוטיקה
ציטוטוקסיקה
צורת מתן:
פומי : PER OS
צורת מינון:
קפסולות בשחרור מושהה : MODIFIED RELEASE CAPSULES
עלון לרופא
מינוניםPosology התוויות
Indications תופעות לוואי
Adverse reactions התוויות נגד
Contraindications אינטראקציות
Interactions מינון יתר
Overdose הריון/הנקה
Pregnancy & Lactation אוכלוסיות מיוחדות
Special populations תכונות פרמקולוגיות
Pharmacological properties מידע רוקחי
Pharmaceutical particulars אזהרת שימוש
Special Warning עלון לרופא
Physicians Leaflet
Adverse reactions : תופעות לוואי
7 ADVERSE REACTIONS The following serious adverse reactions are described below and elsewhere in labeling: • Acute Tubulointerstitial Nephritis [see Warnings and Precautions (6.2)] • Clostridium difficile - Associated Diarrhea [see Warnings and Precautions (6.3)] • Bone Fracture [see Warnings and Precautions (6.4)] • Severe Cutaneous Adverse Reactions [see Warnings and Precautions (6.5)] •Cutaneous and Systemic Lupus Erythematosus [see Warnings and Precautions (6.6)] •Cyanocobalamin (Vitamin B12) Deficiency [see Warnings and Precautions (6.7)] • Hypomagnesemia and Mineral Metabolism [see Warnings and Precautions (6.8)] • Fundic Gland Polyps [see Warnings and Precautions (6.11)] 7.1 Clinical Trials Experience Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. Adults The safety of DEXILANT was evaluated in 4548 adult patients in controlled and single-arm clinical trials, including 863 patients treated for at least six months and 203 patients treated for one year. Patients ranged in age from 18 to 90 years (median age 48 years), with 54% female, 85% Caucasian, 8% Black, 4% Asian, and 3% Other races. Six randomized controlled clinical trials were conducted for the treatment of erosive reflux oesophagitis, maintenance of healed erosive reflux oesophagitis, and symptomatic GORD, which included 896 patients on placebo, 455 patients on DEXILANT 30 mg, 2218 patients on DEXILANT 60 mg, and 1363 patients on lansoprazole 30 mg once daily. Common Adverse Reactions The most common adverse reactions (≥2%) that occurred at a higher incidence for DEXILANT than placebo in the controlled studies are presented in Table 2. Table 2. Common Adverse Reactions in Controlled Studies in Adults Placebo DEXILAN DEXILAN DEXILAN Lansoprazol T T T e Adverse (N=896 30 mg 60 mg Total 30 mg Reaction ) (N=455) (N=2218) (N=2621) (N=1363) % % % % % Diarrhea 2.9 5.1 4.7 4.8 3.2 Abdominal 3.5 3.5 4.0 4.0 2.6 Pain Nausea 2.6 3.3 2.8 2.9 1.8 Upper 0.8 2.9 1.7 1.9 0.8 Respirator y Tract Infection Vomiting 0.8 2.2 1.4 1.6 1.1 Flatulence 0.6 2.6 1.4 1.6 1.2 Adverse Reactions Resulting in Discontinuation In controlled clinical studies, the most common adverse reaction leading to discontinuation from DEXILANT was diarrhea (0.7%). Less Common Adverse Reactions Other adverse reactions that were reported in controlled studies at an incidence of less than 2% are listed below by body system: Blood and Lymphatic System Disorders: anemia, lymphadenopathy Cardiac Disorders: angina, arrhythmia, bradycardia, chest pain, edema, myocardial infarction, palpitation, tachycardia Ear and Labyrinth Disorders: ear pain, tinnitus, vertigo Endocrine Disorders: goiter Eye Disorders: eye irritation, eye swelling Gastrointestinal Disorders: abdominal discomfort, abdominal tenderness, abnormal feces, anal discomfort, Barrett’s esophagus, bezoar, bowel sounds abnormal, breath odor, colitis microscopic, colonic polyp, constipation, dry mouth, duodenitis, dyspepsia, dysphagia, enteritis, eructation, oesophagitis, gastric polyp, gastritis, gastroenteritis, gastrointestinal disorders, gastrointestinal hypermotility disorders, GORD, GI ulcers and perforation, hematemesis, hematochezia, hemorrhoids, impaired gastric emptying, irritable bowel syndrome, mucus stools, oral mucosal blistering, painful defecation, proctitis, paresthesia oral, rectal hemorrhage, retching General Disorders and Administration Site Conditions: adverse drug reaction, asthenia, chest pain, chills, feeling abnormal, inflammation, mucosal inflammation, nodule, pain, pyrexia Hepatobiliary Disorders: biliary colic, cholelithiasis, hepatomegaly Immune System Disorders: hypersensitivity Infections and Infestations: candida infections, influenza, nasopharyngitis, oral herpes, pharyngitis, sinusitis, viral infection, vulvo-vaginal infection Injury, Poisoning and Procedural Complications: falls, fractures, joint sprains, overdose, procedural pain, sunburn Laboratory Investigations: ALP increased, ALT increased, AST increased, bilirubin decreased/increased, blood creatinine increased, blood gastrin increased, blood glucose increased, blood potassium increased, liver function test abnormal, platelet count decreased, total protein increased, weight increase Metabolism and Nutrition Disorders: appetite changes, hypercalcemia, hypokalemia Musculoskeletal and Connective Tissue Disorders: arthralgia, arthritis, muscle cramps, musculoskeletal pain, myalgia Nervous System Disorders: altered taste, convulsion, dizziness, headaches, migraine, memory impairment, paresthesia, psychomotor hyperactivity, tremor, trigeminal neuralgia Psychiatric Disorders: abnormal dreams, anxiety, depression, insomnia, libido changes Renal and Urinary Disorders: dysuria, micturition urgency Reproductive System and Breast Disorders: dysmenorrhea, dyspareunia, menorrhagia, menstrual disorder Respiratory, Thoracic and Mediastinal Disorders: aspiration, asthma, bronchitis, cough, dyspnea, hiccups, hyperventilation, respiratory tract congestion, sore throat Skin and Subcutaneous Tissue Disorders: acne, dermatitis, erythema, pruritus, rash, skin lesion, urticaria Vascular Disorders: deep vein thrombosis, hot flush, hypertension Additional adverse reactions that were reported in a long-term single-arm trial and were considered related to DEXILANT by the treating physician included: anaphylaxis, auditory hallucination, B-cell lymphoma, bursitis, central obesity, cholecystitis acute, dehydration, diabetes mellitus, dysphonia, epistaxis, folliculitis, gout, herpes zoster, hyperlipidemia, hypothyroidism, increased neutrophils, MCHC decrease, neutropenia, rectal tenesmus, restless legs syndrome, somnolence, tonsillitis. Pediatrics The safety of DEXILANT was evaluated in controlled and single-arm clinical trials including 166 pediatric patients, 12 to 17 years of age for the treatment of symptomatic non-erosive GORD, treatment of erosive reflux oesophagitis, maintenance of healed erosive reflux oesophagitis and maintenance of relief of heartburn [see Clinical Studies (14.4)]. The adverse reaction profile was similar to that of adults. The most common adverse reactions that occurred in ≥ 5% of patients were headache, abdominal pain, diarrhea, nasopharyngitis and oropharyngeal pain. Other Adverse Reactions See the full prescribing information for lansoprazole for other adverse reactions not observed with DEXILANT. 7.2 Postmarketing Experience The following adverse reactions have been identified during post-approval use of DEXILANT. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Blood and Lymphatic System Disorders: autoimmune hemolytic anemia, idiopathic thrombocytopenic purpura Ear and Labyrinth Disorders: deafness Eye Disorders: blurred vision Gastrointestinal Disorders: oral edema, pancreatitis, fundic gland polyps General Disorders and Administration Site Conditions: facial edema Hepatobiliary Disorders: drug-induced hepatitis Immune System Disorders: anaphylactic shock (requiring emergency intervention), exfoliative dermatitis, Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) (some fatal), DRESS, AGEP, erythema multiforme. Infections and Infestations: Clostridium difficile associated diarrhea Metabolism and Nutrition Disorders: hypomagnesemia, hypocalcemia, hypokalemia, hyponatremia Musculoskeletal System Disorders: bone fracture Nervous System Disorders: cerebrovascular accident, transient ischemic attack Renal and Genitourinary Disorders: acute renal failure, erectile dysfunction. Respiratory, Thoracic and Mediastinal Disorders: pharyngeal edema, throat tightness Skin and Subcutaneous Tissue Disorders: generalized rash, leukocytoclastic vasculitis Reporting suspected adverse reactions after authorization of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Any suspected adverse events should be reported to the Ministry of Health according to the National Regulation by using an online form https://sideeffects.health.gov.il

שימוש לפי פנקס קופ''ח כללית 1994
לא צוין
תאריך הכללה מקורי בסל
לא צוין
הגבלות
לא צוין
מידע נוסף
