Quest for the right Drug
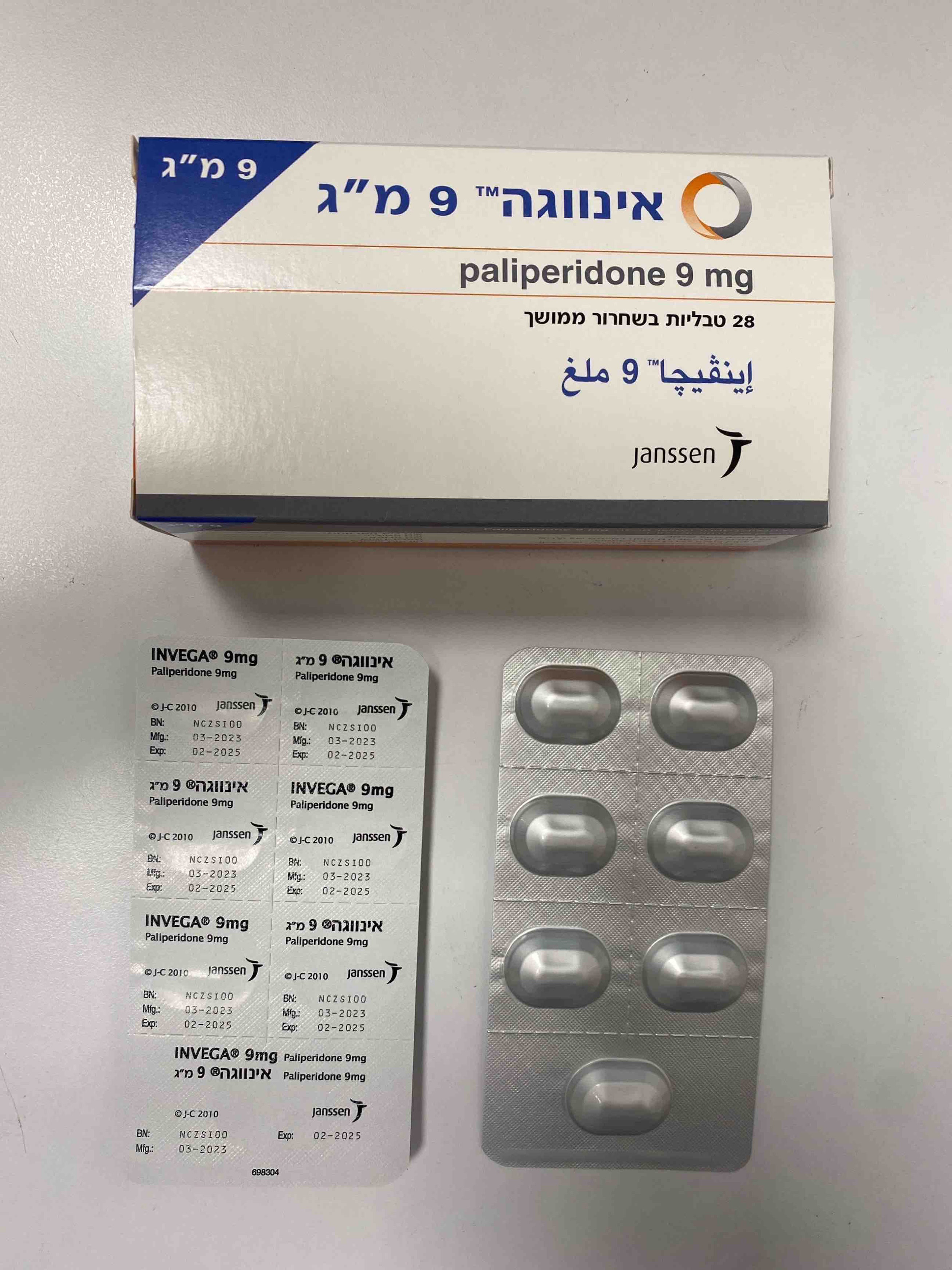
אינווגה 9 מ"ג INVEGA 9 MG (PALIPERIDONE)
תרופה במרשם
תרופה בסל
נרקוטיקה
ציטוטוקסיקה
צורת מתן:
פומי : PER OS
צורת מינון:
טבליות עם שחרור נרחב : TABLETS EXTENDED RELEASE
עלון לרופא
מינוניםPosology התוויות
Indications תופעות לוואי
Adverse reactions התוויות נגד
Contraindications אינטראקציות
Interactions מינון יתר
Overdose הריון/הנקה
Pregnancy & Lactation אוכלוסיות מיוחדות
Special populations תכונות פרמקולוגיות
Pharmacological properties מידע רוקחי
Pharmaceutical particulars אזהרת שימוש
Special Warning עלון לרופא
Physicians Leaflet
Pharmacological properties : תכונות פרמקולוגיות
Pharmacodynamic Properties
5.1 Pharmacodynamic properties Pharmacologic group: Psycholeptics, other antipsychotics ATC code: N05AX13 INVEGA contains a racemic mixture of (+)- and (-)-paliperidone. Mechanism of action Paliperidone is a selective blocking agent of monoamine effects, whose pharmacological properties are different from that of traditional neuroleptics. Paliperidone binds strongly to serotonergic 5-HT2- and dopaminergic D2-receptors. Paliperidone also blocks alfa1-adrenergic receptors and blocks, to a lesser extent, H1-histaminergic and alfa2-adrenergic receptors. The pharmacological activity of the (+)- and (-)-paliperidone enantiomers are qualitatively and quantitatively similar. Paliperidone is not bound to cholinergic receptors. Even though paliperidone is a strong D2-antagonist, which is believed to relieve the positive symptoms of schizophrenia, it causes less catalepsy and decreases motor functions to a lesser extent than traditional neuroleptics. Dominating central serotonin antagonism may reduce the tendency of paliperidone to cause extrapyramidal side effects. Clinical efficacy Invega SPC 9-24 Schizophrenia The efficacy of INVEGA in the treatment of schizophrenia was established in three multi-centre, placebo- controlled, double-blind, 6-week trials in subjects who met DSM-IV criteria for schizophrenia. INVEGA doses, which varied across the three studies, ranged from 3 to 15 mg once daily. The primary efficacy endpoint was defined as a decrease in total Positive and Negative Syndrome Scale (PANSS) scores as shown in the following table. The PANSS is a validated multi-item inventory composed of five factors to evaluate positive symptoms, negative symptoms, disorganised thoughts, uncontrolled hostility/excitement, and anxiety/depression. All tested doses of INVEGA separated from placebo on day 4 (p<0.05). Predefined secondary endpoints included the Personal and Social Performance (PSP) scale and the Clinical Global Impression – Severity (CGI-S) scale. In all three studies, INVEGA was superior to placebo on PSP and CGI-S. Efficacy was also evaluated by calculation of treatment response (defined as decrease in PANSS Total Score ≥ 30%) as a secondary endpoint. Schizophrenia Studies: Positive and Negative Syndrome Scale for Schizophrenia (PANSS) Total Score - Change From Baseline to End Point - LOCF for Studies R076477-SCH-303, R076477-SCH-304, and R076477-SCH-305: Intent-to-Treat Analysis Set Placebo INVEGA INVEGA INVEGA INVEGA 3 mg 6 mg 9 mg 12 mg R076477-SCH-303 (N=126) (N=123) (N=122) (N=129) Mean baseline (SD) 94.1 (10.74) 94.3 (10.48) 93.2 (11.90) 94.6 (10.98) Mean change (SD) -4.1 (23.16) -17.9 (22.23) -17.2 (20.23) -23.3 (20.12) P-value (vs, Placebo) <0.001 <0.001 <0.001 Diff. of LS Means (SE) -13.7 (2.63) -13.5 (2.63) -18.9 (2.60) R076477-SCH-304 (N=105) (N=111) (N=111) Mean baseline (SD) 93.6 (11.71) 92.3 (11.96) 94.1 (11.42) Mean change (SD) -8.0 (21.48) -15.7 (18.89) -17.5 (19.83) P-value (vs, Placebo) 0.006 <0.001 Diff. of LS Means (SE) -7.0 (2.36) -8.5 (2.35) R076477-SCH-305 (N=120) (N=123) (N=123) Mean baseline (SD) 93.9 (12.66) 91.6 (12.19) 93.9 (13.20) Mean change (SD) -2.8 (20.89) -15.0 (19.61) -16.3 (21.81) P-value (vs, Placebo) <0.001 <0.001 Diff. of LS Means (SE) -11.6 (2.35) -12.9 (2.34) Note: Negative change in score indicates improvement. For all 3 studies, an active control (olanzapine at a dose of 10 mg) was included. LOCF = last observation carried forward. The 1-7 version of the PANSS was used. A 15 mg dose was also included in Study R076477-SCH-305, but results are not presented since this is above the maximum recommended daily dose of 12 mg. Schizophrenia Studies: Proportion of Subjects with Responder Status at LOCF End Point Studies R076477-SCH-303, R076477-SCH-304, and R076477-SCH-305: Intent-to-Treat Analysis Set Placebo INVEGA INVEGA INVEGA INVEGA 3 mg 6 mg 9 mg 12 mg R076477-SCH-303 N 126 123 122 129 Responder, n (%) 38 (30.2) 69 (56.1) 62 (50.8) 79 (61.2) Non-responder, n (%) 88 (69.8) 54 (43.9) 60 (49.2) 50 (38.8) P value (vs Placebo) -- <0.001 0.001 <0.001 R076477-SCH-304 N 105 110 111 Responder, n (%) 36 (34.3) 55 (50.0) 57 (51.4) Non-responder, n (%) 69 (65.7) 55 (50.0) 54 (48.6) P value (vs Placebo) -- 0.025 0.012 R076477-SCH-305 N 120 123 123 Responder, n (%) 22 (18.3) 49 (39.8) 56 (45.5) Invega SPC 9-24 Non-responder, n (%) 98 (81.7) 74 (60.2) 67 (54.5) P value (vs Placebo) -- 0.001 <0.001 In a long-term trial designed to assess the maintenance of effect, INVEGA was significantly more effective than placebo in maintaining symptom control and delaying relapse of schizophrenia. After having been treated for an acute episode for 6 weeks and stabilised for an additional 8 weeks with INVEGA (doses ranging from 3 to 15 mg once daily) patients were then randomised in a double-blind manner to either continue on INVEGA or on placebo until they experienced a relapse in schizophrenia symptoms. The trial was stopped early for efficacy reasons by showing a significantly longer time to relapse in patients treated with INVEGA compared to placebo (p=0.0053). Schizoaffective disorder The efficacy of INVEGA in the acute treatment of psychotic or manic symptoms of schizoaffective disorder was established in two placebo-controlled, 6-week trials in non-elderly adult subjects. Enrolled subjects 1) met DSM-IV criteria for schizoaffective disorder, as confirmed by the Structured Clinical Interview for DSM-IV Disorders, 2) had a Positive and Negative Syndrome Scale (PANSS) total score of at least 60, and 3) had prominent mood symptoms as confirmed by a score of at least 16 on the Young Mania Rating Scale (YMRS) and/or Hamilton Rating Scale 21 for Depression (HAM-D 21). The population included subjects with schizoaffective bipolar and depressive types. In one of these trials, efficacy was assessed in 211 subjects who received flexible doses of INVEGA (3-12 mg once daily). In the other study, efficacy was assessed in 203 subjects who were assigned to one of two dose levels of INVEGA: 6 mg with the option to reduce to 3 mg (n = 105) or 12 mg with the option to reduce to 9 mg (n = 98) once daily. Both studies included subjects who received INVEGA either as monotherapy or in combination with mood stabilisers and/or antidepressants. Dosing was in the morning without regard to meals. Efficacy was evaluated using the PANSS. The INVEGA group in the flexible-dose study (dosed between 3 and 12 mg/day, mean modal dose of 8.6 mg/day) and the higher dose group of INVEGA in the 2 dose-level study (12 mg/day with option to reduce to 9 mg/day) were each superior to placebo in the PANSS at 6 weeks. In the lower dose group of the 2 dose- level study (6 mg/day with option to reduce to 3 mg/day), INVEGA was not significantly different from placebo as measured by the PANSS. Only few subjects received the 3 mg dose in both studies and efficacy of this dose could not be established. Statistically superior improvements in manic symptoms as measured by YMRS (secondary efficacy scale) were observed in patients from the flexible-dose study and the INVEGA higher dose in the second study. Taking the results of both studies together (pooled study-data), INVEGA improved the psychotic and manic symptoms of schizoaffective disorder at endpoint relative to placebo when administered either as monotherapy or in combination with mood stabilisers and/or antidepressants. However, overall the magnitude of effect in regard to PANSS and YMRS observed on monotherapy was larger than that observed with concomitant antidepressants and/or mood stabilisers. Moreover, in the pooled population, INVEGA was not efficacious in patients concomitantly receiving mood stabiliser and antidepressants in regard to the psychotic symptoms, but this population was small (30 responders in the paliperidone group and 20 responders in the placebo group). Additionally, in study SCA-3001 in the ITT population the effect on psychotic symptoms measured by PANSS was clearly less pronounced and not reaching statistical significance for patients receiving concomitantly mood stabilisers and/or antidepressants. An effect of INVEGA on depressive symptoms was not demonstrated in these studies, but has been demonstrated in a long-term study with the long-acting injectable formulation of paliperidone (described further down in this section). An examination of population subgroups did not reveal any evidence of differential responsiveness on the basis of gender, age, or geographic region. There were insufficient data to explore differential effects based on race. Efficacy was also evaluated by calculation of treatment response (defined as decrease in PANSS Total Invega SPC 9-24 Score ≥ 30% and CGI-C Score ≤ 2) as a secondary endpoint. Schizoaffective Disorder Studies: Primary Efficacy Parameter, PANSS Total Score Change from Baseline from Studies R076477-SCA-3001 and R076477-SCA-3002: Intent-to-Treat Analysis Set Placebo INVEGA INVEGA Higher INVEGA Flexible Lower Dose Dose Dose (3-12 mg) (3-6 mg) (9-12 mg) R076477-SCA-3001 (N=107) (N=105) (N=98) Mean baseline (SD) 91.6 (12.5) 95.9 (13.0) 92.7 (12.6) Mean change (SD) -21.7 (21.4) -27.4 (22.1) -30.6 (19.1) P-value (vs. Placebo) 0.187 0.003 Diff. of LS Means (SE) -3.6 (2.7) -8.3 (2.8) R076477-SCA-3002 (N=93) (N=211) Mean baseline (SD) 91.7 (12.1) 92.3 (13.5) Mean change (SD) -10.8 (18.7) -20.0 (20.23) P-value (vs. Placebo) <0.001 Diff. of LS Means (SE) -13.5 (2.63) Note: Negative change in score indicates improvement. LOCF = last observation carried forward. Invega SPC 9-24 Schizoaffective Disorder Studies: Secondary Efficacy Parameter, Proportion of Subjects with Responder Status at LOCF End Point: Studies R076477-SCA-3001 and R076477-SCA-3002: Intent-to-Treat Analysis Set Placebo INVEGA INVEGA Higher INVEGA Flexible Lower Dose Dose Dose (3-12 mg) (3-6 mg) (9-12 mg) R076477-SCA-3001 N 107 104 98 Responder, n (%) 43 (40.2) 59 (56.7) 61 (62.2) Non-responder, n (%) 64 (59.8) 45 (43.3) 37 (37.8) P value (vs Placebo) -- 0.008 0.001 R076477-SCA-3002 N 93 210 Responder, n (%) 26 (28.0) 85 (40.5) Non-responder, n (%) 67 (72.0) 125 (59.5) P value (vs Placebo) -- 0.046 Response defined as decrease from baseline in PANSS Total Score ≥ 30% and CGI-C Score ≤ 2 In a long-term trial designed to assess the maintenance of effect, the long-acting injectable formulation of paliperidone was significantly more effective than placebo in maintaining symptom control and delaying relapse of psychotic, manic, and depressive symptoms of schizoaffective disorder. After having been successfully treated for an acute psychotic or mood episode for 13 weeks and stabilised for an additional 12 weeks with the long-acting injectable formulation of paliperidone (doses ranging from 50 to 150 mg) patients were then randomised to a 15-month double-blind relapse prevention period of the study to either continue on the long-acting injectable formulation of paliperidone or on placebo until they experienced a relapse of schizoaffective symptoms. The study showed a significantly longer time to relapse in patients treated with the long-acting injectable formulation of paliperidone compared to placebo (p<0.001). Paediatric population See section 4.2 for information on paediatric use. The efficacy of INVEGA in adolescent subjects with schizophrenia (INVEGA N = 149, placebo N = 51) was studied in a randomised, double-blind, placebo-controlled, 6-week study using a fixed-dose weight-based treatment group design over the dose range of 1.5 mg/day to 12 mg/day. Subjects were 12-17 years of age and met DSM-IV criteria for schizophrenia. Efficacy was evaluated using PANSS. This study demonstrated the efficacy of INVEGA of the medium dose group in adolescent subjects with schizophrenia. Secondary by dose analysis demonstrated the efficacy of 3 mg, 6 mg, and 12 mg dose given once daily. Adolescent Schizophrenia Study: R076477-PSZ-3001: 6-week, fixed-dose, placebo-controlled Intent-to-Treat Analysis Set. LOCF endpoint change from baseline Placebo INVEGA INVEGA INVEGA Low Dose Medium Dose High Dose 1.5 mg 3 or 6 mg* 6 or 12 mg** N=51 N=54 N=48 N=47 Change in PANSS Score Mean baseline (SD) 90.6 (12.13) 91.6 (12.54) 90.6 (14.01) 91.5 (13.86) Mean change (SD) -7.9 (20.15) -9.8 (16.31) -17.3 (14.33) -13.8 (15.74) P-value (vs Placebo) 0.508 0.006 0.086 Diff. of LS Means (SE) -2.1 (3.17) -10.1 (3.27) -6.6 (3.29) Invega SPC 9-24 Responder Analysis Responder, n (%) 17 (33.3) 21 (38.9) 31 (64.6) 24 (51.1) Non-responder, n (%) 34 (66.7) 33 (61.1) 17 (35.4) 23 (48.9) P value (vs Placebo) 0.479 0.001 0.043 Response defined as decrease from baseline in PANSS Total Score ≥ 20% Note: Negative change in score indicates improvement. LOCF = last observation carried forward. * Medium dose group: 3 mg for subjects < 51 kg, 6 mg for subjects ≥ 51 kg ** High dose group: 6 mg for subjects < 51 kg, 12 mg for subjects ≥ 51 kg Efficacy of INVEGA over a flexible dose range of 3 mg/day to 9 mg/day in adolescent subjects (12 years and older) with schizophrenia (INVEGA N = 112, aripiprazole N = 114) was also evaluated in a randomised, double-blind, active-controlled study that included an 8-week, double-blind acute phase and an 18-week, double-blind maintenance phase. The changes in PANSS total scores from baseline to Week 8 and Week 26 were numerically similar between the INVEGA and aripiprazole treatment groups. In addition, the difference in the percentage of patients demonstrating ≥ 20% improvement in PANSS total score at Week 26 between the two treatment groups was numerically similar. Adolescent Schizophrenia Study: R076477-PSZ-3003: 26-week, flexible-dose, active-controlled Intent-to-Treat Analysis Set. LOCF endpoint change from baseline INVEGA Aripiprazole 3-9 mg 5-15 mg N=112 N=114 Change in PANSS Score 8 week, acute endpoint Mean baseline (SD) 89.6 (12.22) 92.0 (12.09) Mean change (SD) -19.3 (13.80) -19.8 (14.56) P-value (vs aripiprazole) 0.935 Diff. of LS Means (SE) 0.1 (1.83) Change in PANSS Score 26 week endpoint Mean baseline (SD) 89.6 (12.22) 92.0 (12.09) Mean change (SD) -25.6 (16.88) -26.8 (18.82) P-value (vs aripiprazole) 0.877 Diff. of LS Means (SE) -0.3 (2.20) Responder Analysis 26 week endpoint Responder, n (%) 86 (76.8) 93 (81.6) Non-responder, n (%) 26 (23.2) 21 (18.4) P value (vs aripiprazole) 0.444 Response defined as decrease from baseline in PANSS Total Score ≥ 20% Note: Negative change in score indicates improvement. LOCF = last observation carried forward.
Pharmacokinetic Properties
5.2 Pharmacokinetic properties The pharmacokinetics of paliperidone following INVEGA administration are dose proportional within the available dose range. Absorption Following a single dose, INVEGA exhibits a gradual ascending release rate, allowing the plasma concentrations of paliperidone to steadily rise to reach peak plasma concentration (Cmax) approximately 24 hours after dosing. With once-daily dosing of INVEGA, steady-state concentrations of paliperidone are attained within 4-5 days of dosing in most subjects. Paliperidone is the active metabolite of risperidone. The release characteristics of INVEGA result in minimal INVEGA SPC 9-24 peak-trough fluctuations as compared to those observed with immediate-release risperidone (fluctuation index 38% versus 125%). The absolute oral bioavailability of paliperidone following INVEGA administration is 28% (90% CI of 23%- 33%). Administration of paliperidone extended-release tablets with a standard high-fat/high-caloric meal increases Cmax and AUC of paliperidone by up to 50-60% compared with administration in the fasting state. Distribution Paliperidone is rapidly distributed. The apparent volume of distribution is 487 l. The plasma protein binding of paliperidone is 74%. It binds primarily to α1-acid glycoprotein and albumin. Biotransformation and elimination One week following administration of a single oral dose of 1 mg immediate-release 14C-paliperidone, 59% of the dose was excreted unchanged into urine, indicating that paliperidone is not extensively metabolised by the liver. Approximately 80% of the administered radioactivity was recovered in urine and 11% in the faeces. Four metabolic pathways have been identified in vivo, none of which accounted for more than 6.5% of the dose: dealkylation, hydroxylation, dehydrogenation, and benzisoxazole scission. Although in vitro studies suggested a role for CYP2D6 and CYP3A4 in the metabolism of paliperidone, there is no evidence in vivo that these isozymes play a significant role in the metabolism of paliperidone. Population pharmacokinetics analyses indicated no discernible difference on the apparent clearance of paliperidone after administration of INVEGA between extensive metabolisers and poor metabolisers of CYP2D6 substrates. In vitro studies in human liver microsomes showed that paliperidone does not substantially inhibit the metabolism of medicines metabolised by cytochrome P450 isozymes, including CYP1A2, CYP2A6, CYP2C8/9/10, CYP2D6, CYP2E1, CYP3A4, and CYP3A5. The terminal elimination half-life of paliperidone is about 23 hours. In vitro studies have shown that paliperidone is a P-gp substrate and a weak inhibitor of P-gp at high concentrations. No in vivo data are available and the clinical relevance is unknown. Hepatic impairment Paliperidone is not extensively metabolised in the liver. In a study in subjects with moderate hepatic impairment (Child-Pugh class B), the plasma concentrations of free paliperidone were similar to those of healthy subjects. No data are available in patients with severe hepatic impairment (Child-Pugh class C). Renal impairment Elimination of paliperidone decreased with decreasing renal function. Total clearance of paliperidone was reduced in subjects with impaired renal function by 32% in mild (Creatinine Clearance [Cr Cl] = 50 to < 80 ml/min), 64% in moderate (CrCl = 30 to < 50 ml/min), and 71% in severe (CrCl = < 30 ml/min) renal impairment. The mean terminal elimination half-life of paliperidone was 24, 40, and 51 hours in subjects with mild, moderate, and severe renal impairment, respectively, compared with 23 hours in subjects with normal renal function (CrCl ≥ 80 ml/min). Elderly Data from a pharmacokinetic study in elderly subjects (≥ 65 years of age, n = 26) indicated that the apparent steady-state clearance of paliperidone following INVEGA administration was 20% lower compared to that of adult subjects (18-45 years of age, n = 28). However, there was no discernable effect of age in the population pharmacokinetic analysis involving schizophrenia subjects after correction of age-related decreases in CrCl. Adolescents INVEGA SPC 9-24 Paliperidone systemic exposure in adolescent subjects (15 years and older) was comparable to that in adults. In adolescents weighing < 51 kg, a 23% higher exposure was observed than in adolescents weighing ≥ 51 kg. Age alone did not influence the paliperidone exposure. Race Population pharmacokinetics analysis revealed no evidence of race-related differences in the pharmacokinetics of paliperidone following INVEGA administration. Gender The apparent clearance of paliperidone following INVEGA administration is approximately 19% lower in women than men. This difference is largely explained by differences in lean body mass and creatinine clearance between men and women. Smoking status Based on in vitro studies utilising human liver enzymes, paliperidone is not a substrate for CYP1A2; smoking should, therefore, not have an effect on the pharmacokinetics of paliperidone. A population pharmacokinetic analysis showed a slightly lower exposure to paliperidone in smokers compared with non-smokers. The difference is unlikely to be of clinical relevance, though.

פרטי מסגרת הכללה בסל
1. הטיפול בתרופה האמורה יינתן לאחד מאלה: א. למבוטח בגיר שהוא חולה סכיזופרניה, ובהתקיים אחד מהתנאים האלה: 1. המטופל מוגדר כבעל קווי התנהגות תוקפניים, וכטיפול ראשון; 2. המטופל לא הגיב לטיפול בתרופה אנטי פסיכוטית אטיפית שניתנה לו כקו טיפול ראשון, או פיתח תופעות לוואי קשות לטיפול כאמור; ב. למבוטח קטין הסובל מסכיזופרניה או מפסיכוזה אחרת, וכטיפול ראשון; 2. התחלת הטיפול בתרופה תהיה על פי הוראתו של רופא מומחה בפסיכיאטריה או בפסיכיאטריה של הילד והמתבגר או בנוירולוגיה, לפי העניין. 3. לא יינתנו לחולה בו בזמן שתי תרופות או יותר ממשפחת התרופות האנטיפסיכוטיות האטיפיות.
מסגרת הכללה בסל
התוויות הכלולות במסגרת הסל
| התוויה | תאריך הכללה | תחום קליני | Class Effect | מצב מחלה |
|---|---|---|---|---|
| למבוטח קטין הסובל מסכיזופרניה או מפסיכוזה אחרת; | ||||
| למבוטח בגיר שהוא חולה סכיזופרניה; |
שימוש לפי פנקס קופ''ח כללית 1994
לא צוין
תאריך הכללה מקורי בסל
01/01/2009
הגבלות
תרופה מוגבלת לרישום ע'י רופא מומחה או הגבלה אחרת
מידע נוסף
עלון מידע לצרכן
01.06.22 - עלון לצרכן אנגלית 01.06.22 - עלון לצרכן עברית 01.06.22 - עלון לצרכן ערבית 28.02.23 - עלון לצרכן אנגלית 28.02.23 - עלון לצרכן עברית 28.02.23 - עלון לצרכן ערבית 26.09.24 - עלון לצרכן עברית 07.11.24 - עלון לצרכן אנגלית 07.11.24 - עלון לצרכן עברית 07.11.24 - עלון לצרכן ערבית 30.12.13 - החמרה לעלון 26.09.21 - החמרה לעלון 01.06.22 - החמרה לעלון 26.09.24 - החמרה לעלון 07.11.24 - החמרה לעלוןלתרופה במאגר משרד הבריאות
אינווגה 9 מ"ג