Quest for the right Drug
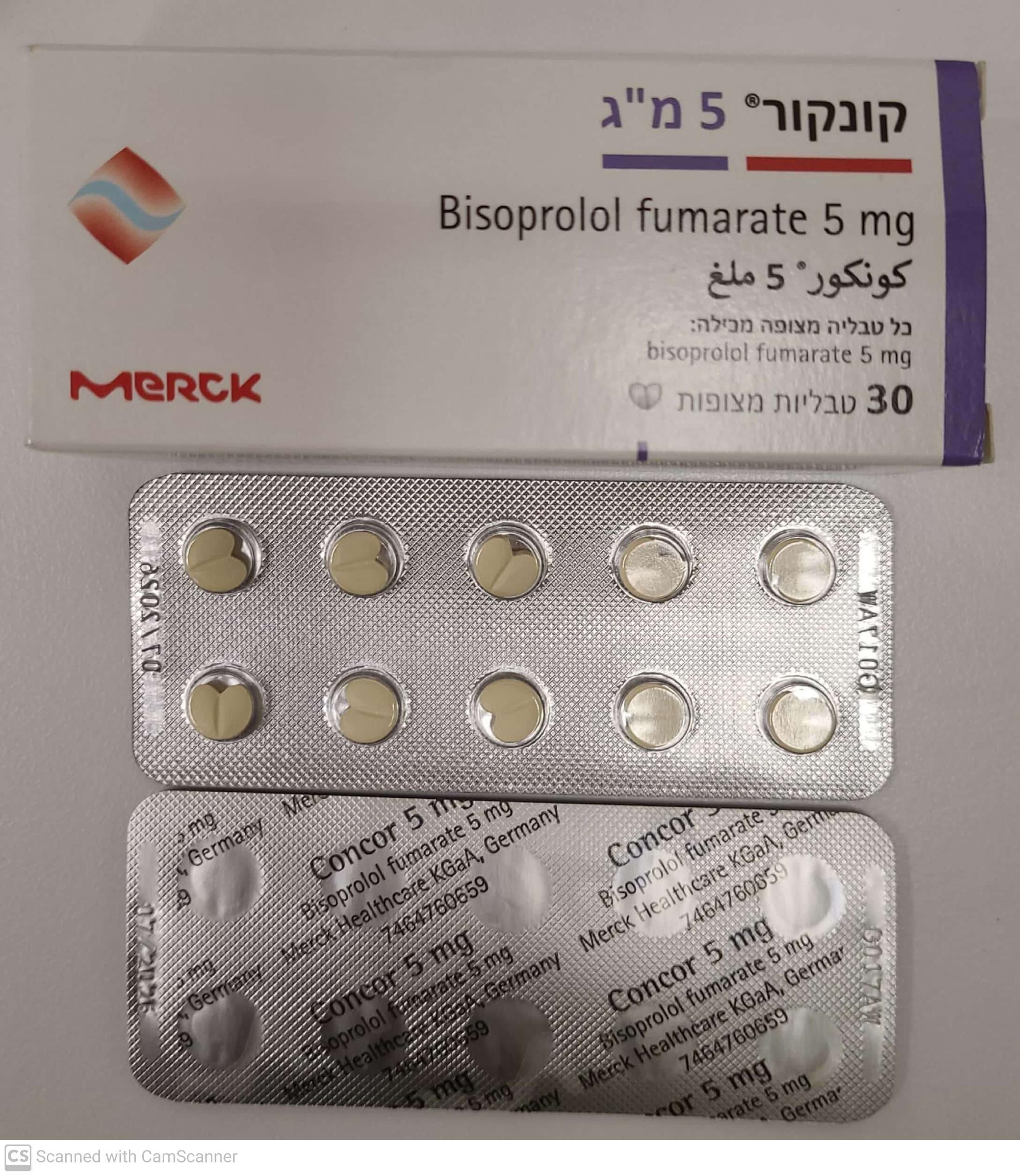
קונקור 5 מ"ג CONCOR 5 MG (BISOPROLOL FUMARATE)
תרופה במרשם
תרופה בסל
נרקוטיקה
ציטוטוקסיקה
צורת מתן:
פומי : PER OS
צורת מינון:
טבליה : TABLETS
עלון לרופא
מינוניםPosology התוויות
Indications תופעות לוואי
Adverse reactions התוויות נגד
Contraindications אינטראקציות
Interactions מינון יתר
Overdose הריון/הנקה
Pregnancy & Lactation אוכלוסיות מיוחדות
Special populations תכונות פרמקולוגיות
Pharmacological properties מידע רוקחי
Pharmaceutical particulars אזהרת שימוש
Special Warning עלון לרופא
Physicians Leaflet
Adverse reactions : תופעות לוואי
4.8 Undesirable effects The following definitions apply to the frequency terminology used hereafter: Very common (≥ 1/10) Common (≥1/100 to <1/10) Uncommon (≥1/1,000 to <1/100) Rare (≥1/10,000 to <1/1,000) Very rare (<1/10,000) Frequency not known (cannot be estimated from available data) Cardiac disorders: Very common: bradycardia. Common: worsening of heart failure. Uncommon: AV-conduction disturbances. Investigations: Rare: increased triglycerides, increased liver enzymes (ALAT, ASAT). Nervous system disorders: Common: dizziness, headache. Rare: syncope. Eye disorders: Rare: reduced tear flow (to be considered if the patient uses lenses). Very rare: conjunctivitis. Ear and labyrinth disorders: Rare: hearing disorders. Respiratory, thoracic and mediastinal disorders: Uncommon: bronchospasm in patients with bronchial asthma or a history of obstructive airways disease. Rare: allergic rhinitis. Gastrointestinal disorders: Common: gastrointestinal complaints such as nausea, vomiting, diarrhoea, constipation. Skin and subcutaneous tissue disorders: Rare: hypersensitivity reactions (pruritus, flush, rash and angioedema). Very rare: alopecia. Beta-blockers may provoke or worsen psoriasis or induce psoriasis-like rash. Musculoskeletal and connective tissue disorders: Uncommon: muscular weakness and cramps. Vascular disorders: Common: feeling of coldness or numbness in the extremities, hypotension. Uncommon: orthostatic hypotension. General disorders: Common: asthenia, fatigue. Hepatobiliary disorders: Rare: hepatitis. Reproductive system and breast disorders: Rare: erectile dysfunction. Psychiatric disorders: Uncommon: sleep disorder, depression. Rare: nightmare, hallucination. Reporting of suspected adverse reactions Reporting suspected adverse reactions after authorisation of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Any suspected adverse events should be reported to the Ministry of Health according to the National Regulation by using an online form https://sideeffects.health.gov.il/

שימוש לפי פנקס קופ''ח כללית 1994
לא צוין
תאריך הכללה מקורי בסל
לא צוין
הגבלות
לא צוין
מידע נוסף
